|
2 The 4 Technology Solutions

REDOX REACTIONS IN REMEDIATION
An On-Line Version of a Column First Published in:
Environmental Technology July/Aug. 1996 Vol. 6 No. 4 Pages 24-25
by: David B. Vance dbv7@mindspring.com
Oxidation and reduction (Redox) reactions play an important role in the geochemical processes that occur in groundwater. Redox reactions are defined as reactions in which electrons are transferred. The species receiving electrons is reduced, that donating electrons is oxidized. Redox reactions determine the mobility of many inorganic compounds as well as biologically important materials such as nitrogen and sulfur. In addition, redox conditions govern the particulars for the biological degradation of complex hydrocarbon contaminants. The purpose of this column is to examine the concept of redox, how redox potential is measured, and its usefulness in the remediation process.
Redox potential is a an intensity parameter of overall redox reaction potential in the system (similar in concept to pH), not the capacity of the system for specific oxidation or reduction reactions. The redox value of standard half reactions (Eo) and details of how to calculate redox capacity can be found in any elementary chemistry text.
In the literature redox potential is generally reported as Eh, which is the potential generated between a platinum electrode and a standard hydrogen electrode when placed into the groundwater being tested, where hydrogen is considered the reference electrode. Measurement of redox potential in groundwater can be a difficult task. The common platinum electrode is not responsive to many reactions involving solid phases, nor to the following soluble phase redox couples that are common in groundwater:
- O2--H2O
- SO42----H2S
- CO2--CH4
- NO3---N2
- N2--NH4+
Most work with redox reactions involves laboratory evaluation under static conditions, the focus is on solutions in closed systems that are in an equilibrium state. In groundwater with more than one redox couple the redox potential can not be accurately measured unless the redox reactants are at thermodynamic equilibrium. However, environmental processes are typically in dis-equilibrium, caused by:
- Flowing groundwater conditions.
- Biological activity.
- Redox reactions of light bioactive elements (C, H, O, and S) which most often involve the breaking of covalent bonds, a process that is normally slow.
- Electrochemical reactions of exposed active mineral surfaces.
Some redox reactions can take from 10 to 1000 years to reach equilibrium.
In oxidizing waters measurement of Eh with a platinum electrode can seldom be correlated to a single specific redox couple, and as such is usually of little value in quantifying the redox chemistry. Groundwater with reducing character can be more accurately tested with the platinum electrode, however, samples of strongly reduced water will have skewed redox values if exposed to air during sampling or testing. The latter places a particular burden on how to purge and sample wells in a manner to insure that the sample is truly indicative of the formation beyond the influence of the well bore.
Instead of measuring the redox potential from a field sample, the redox potential can be calculated using the relative concentrations and redox potential of major dissolved species. This is precisely the process used in fate and transport models utilized in support of risk assessments.
Groundwater typically contains both reduced and oxidized species, however what is termed the redox buffer capacity of an aquifer is normally dominated by the chemistry of the aquifer matrix.
- The oxidation capacity of the aquifer acts as a buffer to limit the transport of a plume in a reduced state that would be generated from hydrocarbon contamination or a landfill.
- The reduction capacity of an aquifer will buffer against attempts to increase the redox potential as part of the remediation program (such as air sparging or in-situ bioremediation) to address a contaminant plume.
- In most natural groundwater systems iron is the dominant (by 1 to 3 orders of magnitude) buffering species involved in redox reactions. In some organic rich aquifers TOC can equal ferrous iron as a reduction buffer.
The following inorganic oxidation reactions will consume dissolved oxygen in groundwater:
- Sulfide Oxidation
2O2 + HS- = SO42- + H+
- Iron Oxidation
O2 + 4Fe+2 + 4H+ = 4Fe3+ + 2H2O
- Nitrification
2O2 + NH4+ = NO3- + 2H+ + H2O
- Manganese (II) Oxidation
O2 + 2Mn2+ + 2H2O = 2MnO2 + 4H+
- Iron Sulfide Oxidation
15O2 + 4FeS2 + 14H2O = 4Fe(OH)3 + 8SO42- + 16H+
The following redox reactions will consume organic matter in groundwater:
- (1) Aerobic Degradation
CH2O + O2 = CO2 + H2O
- (2) Denitrification
5CH2O + 4NO3- = 2N2 + 5HCO3- + H+ + 2H2O
- (3) Manganese (IV) Reduction
CH2O + 2MnO2 + 3H+ = 2Mn2+ + HCO3-+ 2H2O
- (4) Ferric Iron Reduction
CH2O + 4Fe(OH)3 + 7H+ = 4Fe2+ + HCO3- + 10H2O
- (5) Sulfate Reduction
2CH2O + SO42- = HS- + 2HCO3- + H+
- (6) Methane Fermentation
2CH2O + H2O = CH4 + HCO3- + H+
The above reactions are listed in order of decreasing Eh conditions as follows:
- (1) Aerobic Degradation
- (2) Oxygen - Nitrogen
- (3 & 4) Iron - Mn (IV)
- (5) Sulfate
- (6) Methane - Hydrogen
Water in contact with air will have an Eh in the range of 350mv to 500mv. Microbially mediated redox processes may decrease the redox potential to values as low as -300mv.
A method of handling Eh data that has proven of particular value to geochemist is the Eh-pH diagram, on which species oxidation state is plotted with variable Eh and pH conditions. The Eh- pH diagram works well in a qualitative fashion, but due to conditions previously discussed are usually unsuccessful when applied to quantitative applications. The figure illustrates the geologically important iron/water system. In addition to illustrating iron oxidation state, it shows how Eh in groundwater is governed at the upper range by the oxidation of water to O2 and the lower range by the reduction of hydrogen ions to H2.
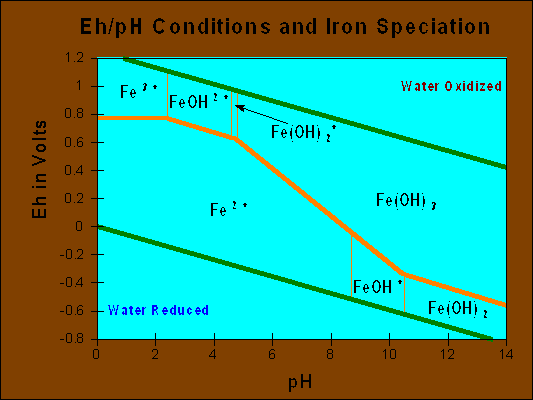
In conclusion, a cautionary note and suggestions for the obtaining the maximum value form the use of redox potential. The caution concerns the field measurement of Eh, it can be difficult to obtain representative samples and Eh measurements themselves may be unrepresentative of true Eh conditions due to the presence of multiple redox couples or general dis-equilibrium. The value of Eh resides primarily an indicator of contaminant transport and as a tool in support of intrinsic remediation activity within a contaminated aquifer. Evaluation for dissolved species generated from reactions 1 through 6 above provides direct evidence of Eh conditions as well as hydrocarbon degradation. Published Eh-pH diagrams can then be used in support of intrinsic remediation arguments, an important tool for those presenting those arguments to the regulatory community.
Return to the Environmental Technology Index Page
Copyright 2013 David B. Vance
All Rights Reserved

If you have comments or suggestions, e-mail me at dbv7@mindspring.com
|