2 The 4 Technology Solutions
![]()
BIOFOULING OF WELLS - CAUSES AND SOLUTIONS
An On-Line Version of a Column First Published in:
Environmental Technology Sept./Oct. 1998 Vol. 8 No. 5
By: David B. Vance dbv7@mindspring.com
Well fouling can occur through physical, chemical, or biological processes. Biological processes are estimated to be associated with 80% of well plugging events. The focus of this column is on mechanisms, diagnosis, and treatment of biofouling.
Symptoms of the fouling of well screens or the adjacent formation include:
- Increased drawdown during pumping of groundwater recovery wells, or increased pressure or required duration for the injection of fluids.
- Reduced water quality.
- Higher iron or manganese concentrations.
- Increased bacteria counts.
Fouling through biological activity can occur due to four primary mechanisms, in order of importance they are:
- The physical presence of extracellular slimes that are excreted by bacteria to aid in their attachment to surfaces within the well bore or formation.
- The accumulation of soil particles and precipitated minerals within those slime layers. Some minerals are collected from the soluble phase by extracellular chemicals excreted by bacteria for that specific purpose.
- Occlusion due to gas generation from active bacteria.
- Corrosion of well components associated with biological activity generating hydrogen sulfide or organic acids.
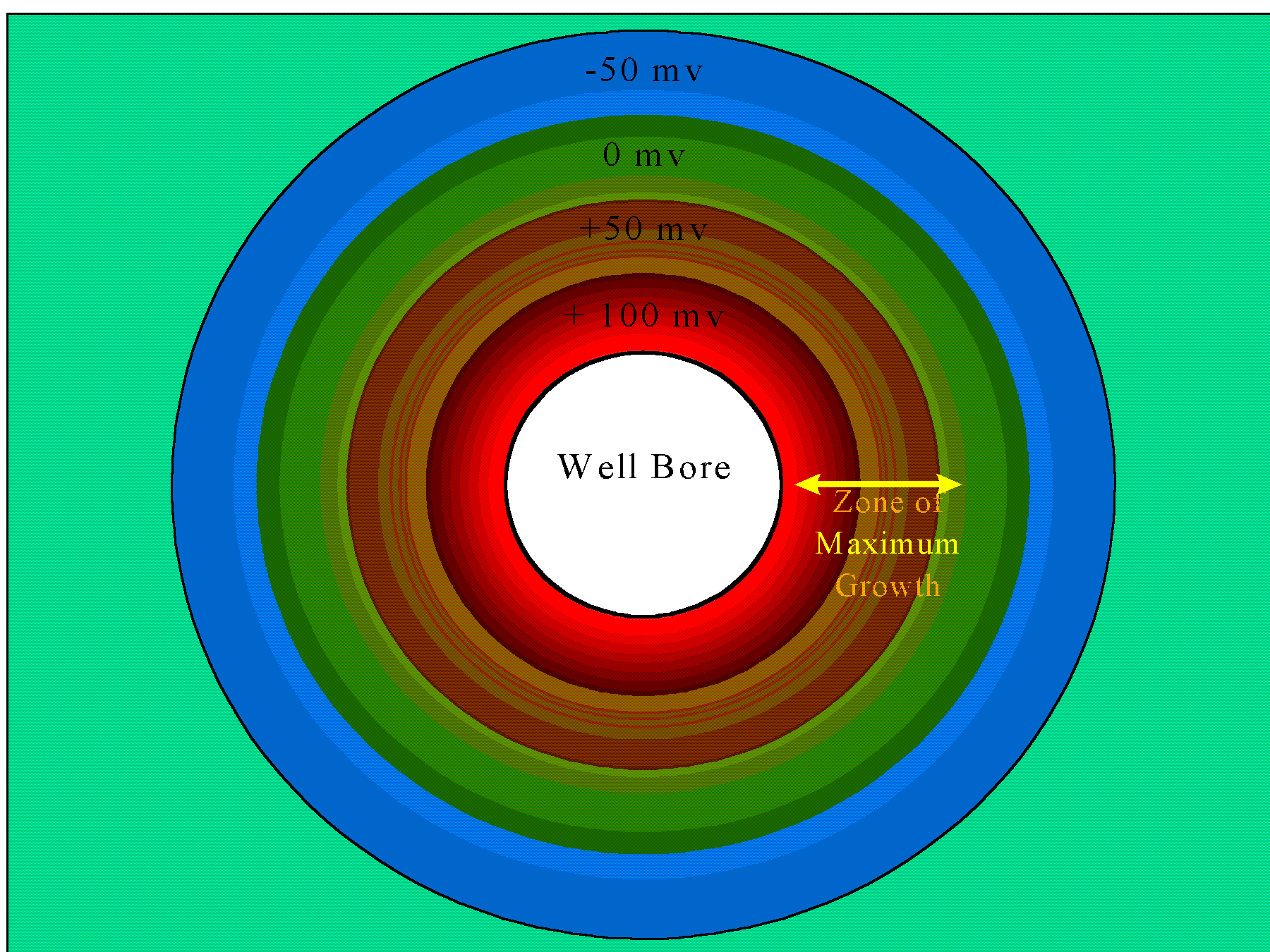
Extracellular slimes are largely composed of polysaccharides and in general are the major component of the biofouling mass. Biomass associated with viable active cells is a relatively minor component. The figure illustrates how the maximum level of biological activity commonly occurs under redox conditions that are at the periphery of oxidative in the Eh range of -50 to +150 mv.
Biofouling events can be complex and caused by a variety of bacteria. However the appearance and odor of the bacterial slimes is diagnostic.
Appearance
- White or clears slime has not accumulated any precipitated minerals which contribute coloration.
- Black slime is the result of sulfate reducing bacteria, soluble minerals are precipitated as black sulfides.
- Grey slime is intermediate with encapsulated black granules or layers that are due to the activity of sulfate reducing bacteria. However, the system is not totally dominated by sulfate reducers.
- Orange, red, pink or brown slime occurs where conditions are significantly or even borderline aerobic. The coloration is from iron and manganese precipitated as oxides or hydroxides within the slime layer or as concretions surrounding active cells.
Odor
- A rotten egg smell is associated with hydrogen sulfide and sulfate reducing bacteria.
- A fish smell is associated with the aerobic growth of pseudomonous species.
- Earth smells are due to a specific class of biologically generated chemicals called geosmins which are associated with streptomycetes or cyanobacteria and are the result of aerobic activity.
- Sewage smell indicates impact from sewage and colliform bacteria.
- Vegetable smells, either fresh or rotting are indicative of green algae or diatoms.
Treatment of well fouling falls into four broad areas:
- Mechanical agitation by surging, water jetting, scrubbing, or air sparging the well bore.
- Acidification to solublize minerals as well as the polysaccharide portion of biological slimes. Acid reacts with the polysaccharides to create simple soluble sugars .
- Flushing to remove suspended or soluble debris that have been physically removed by mechanical agitation.
- The use of bactericides to suppress bacterial populations.
Acidification is the chemical treatment most often applied to plugged wells. It is effective, cheap, and easily applied. In addition to the solubilization of extracellular polysaccharides and precipitated mineral species, it is also bactericidal in its action. The three most commonly used acids are hydrochloric (HCl), sulfamic (H3NO3S), and hydroxyacetic (C2H4O3).
Hydrochloric acid is readily available as muratic acid at a strength of 28 to 31 percent. It should be added into a well at a volume equal to the screened interval of the well bore plus another 25 to 50%. Hydrochloric acid is an extremely effective well cleaning agent, however it does have several drawbacks: it can be corrosive to metal well screens, casings, and pump components; it is also extremely toxic and care should be taken to avoid vapors from the concentrated acid as well a vapors that may be expelled from the well bore during and after acid addition.
Sulfamic acid is used in a solid form and can be introduced into a well bore in a pelletized or granular form. It is most often used to create a 30% solution. It is most effective for treating mineral encrustations that are due to calcium of magnesium. It is less effective for iron or manganese precipitates. Although, the addition of rock salt at a dosage rate of 20% of the acid can aid in the treatment of iron and manganese encrustations. While this material should be handled with the same caution as hydrochloric acid, its potential for fume generation is significantly less. It is also relatively non-corrosive. Sulfamic acid should not be confused with sulfuric acid. Sulfuric acid is corrosive and in my experience more dangerous to deal with than hydrochloric acid. Sulfuric acid will also form insoluble calcium sulfate precipitates within the well bore exacerbating the fouling problem.
Hydroxyacetic acid is also known as glycolic acid and is commonly available as a 70% concentrate. Hydroxyacetic acid can dissolve mineral scale, act as a bactericide, and acts as a chelating agent keeping solublized minerals in solution through the entire treatment process. Treatment dosage is 1 gallon of 70% hydroxyacetic acid for every 10 to 15 gallons of water in the screened interval of the well bore.
In conclusion, it is unlikely that a single anti-fouling procedure will work every time in every well (although acid treatment comes the closest). Seasonal variations will impact the type of biofouling activity in any given well at any given site, much less the variations between different wells or sites. Injection wells in particular have a limited life span that may range from a few years to months. Restoration of the well bore and the adjacent formation is a process that will have limits, a point will be reached where it will become more economic to install a new well near the old.
Return to the Environmental Technology Index Page
Copyright 2008 David B. Vance
All Rights Reserved
![]()
If you have comments or suggestions, email me at dbv7@mindspring.com