|
2 The 4 Technology Solutions

THE UNIVERSAL VIGOR OF MICROBIOLOGICAL SYSTEMS
An On-Line Version of a Column First Published in:
Environmental Technology Sept./Oct. 1996 Vol. 6 No. 5 Pages 28-29
by: David B. Vance dbv7@mindspring.com
This Column was begun the day after it was announced that there is a high probability that microbial life existed on Mars some 3.5 billion years ago. If true, it is a profound event. The focus of this Groundwater Column is on the extreme diversity of microbial life with regards to ambient conditions, metabolic pathways, and geographic dispersion at depth in soils and sedimentary formations, and it appears even on Mars.
First, a little historical perspective is worthwhile with regards to Earth's microbial life:
- From 3.5 to 1.5 billion years ago the atmosphere of the earth was anoxic and the dominant life forms were anaerobic microbes.
- Starting 3.0 billion years ago photosynthetic species began to become active and excrete oxygen as a waste product of their metabolism.
- Between 1.5 and 1.0 billion years ago the "first" great environmental disaster occurred when photosynthetic life became abundant enough to elevate atmospheric oxygen to near current levels.
The type of bacteria that are indicated to have existed on Mars appear to be similar to those on Earth termed archaebacteria. These are bacteria with an ancient history, first appearing on Earth 3.5 billion years ago under the atmospheric conditions described above. Recent advances in genetic analysis indicate that these bacteria are a third form of life different from regular bacteria (Eubacteria) and all multi-cellular life (Eukaryotes). These archaebacteria survive through exotic metabolic processes that rely of a wide variety of chemical reactionsto provide energy.
The archaebacteria fall into four major classes:
- Acidophiles which are able to grow in the pH range of 0.0 to 5.0.
- Thermophiles that can grow in a temperature range of +30 to +250oC.
- Halophiles that survive in 10 to 27% sodium chloride solutions.
- Methanogens which are the best known archaebacteria and form methane in sediments and the intestinal tracts of animals.
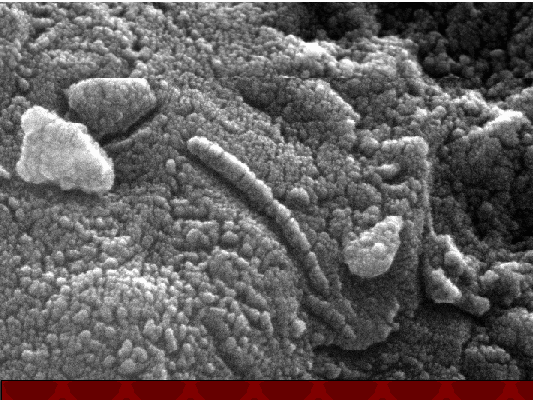
Martian Archaeacteria ?
There are also modern (relatively speaking) Eubacteria that are capable of adaptation to extreme environments including:
- Alkalophiles that can grow in the pH range of 8.5 to 11.5
- Barotolerant bacteria can exist under hydrostatic pressures up to 1,100 atmospheres.
- Other chemoautotrophic bacteria such as denitrifiers, nitrate reducers, ferric iron reducers, sulfate reducers, hydrogen formers, sulfide oxidizers, ferrous iron oxidizers, and still others.
The biochemical diversity is astounding. Just a few specifics are as follow:
- Bacteria have been found in aquifers at depths of 1.5 miles.
- They have been found in oil production formations at depths of almost 4 miles.
- Within the last year bacteria have been found that survive utilizing only surrounding rock and that generate hydrogen gas in the process, these are near 1 mile deep in basalt.
Sulfate reduction is extremely common place in groundwater. Well water with the odor of hydrogen sulfide (rotten eggs) is a manifestation of bacteria driven sulfate reduction.
Additional aquifer conditions under which it is common to find sulfate reducing bacterial species are:
- Eh conditions less than -200 mV and
- the presence of greater than 20 millimolar sulfate from geologic origins.
In the western United States many aquifers are anaerobic and contain sulfate. Anthranilic acid, benzyl alcohol, and P-cresol have all been demonstrated to be biodegraded in the presence of sulfate reducing bacteria. Research is currently under way evaluating the use of sulfate reducers for the degradation of chlorinated solvents and PCBs.
Two other dominant and effective anaerobic processes are denitrification and methanogenesis In general, most aromatic hydrocarbons can be degraded anaerobically under methanogenic or denitrifying conditions.
Some thought should always be put into the exploitation of these alternate bacterial systems. The exact stoichiometrics should always be worked out with a complete assessment of all by-products. For example it is important to evaluate the overall electron flux in nitrate reduction systems:
- For each mole of nitrate consumed one mole of KOH or NaOH is generated.
- This in turn can lower aquifer pH and form limestone or other precipitates, plugging the infiltration or treatment zones.
- An acid must be added concurrent with the nitrate source in the appropriate amount.
Bacteria that produce hydrogen can be used to sequentially dehalogenate chlorinated hydrocarbons to innocuous ethane or ethene gas.
Of course all of these various metabolic systems are not equal with regards to energy yielded to the organism for each reaction that takes place.
On a scale of 1 to 10, with 10 having the highest energy yield:
- Aerobic oxygen based metabolism is 10.
- Nitrate reduction is 9.
- Sulfate reduction is 4.
- Methanogenesis is 2.
Thus the source of the dominance of aerobic life that was precipitated by events one billion years ago (as described above). Given surface conditions it is the most cost effective metabolic system available. However, all of the other metabolic options still take precedent under the appropriate conditions. It has proven particularly difficult to drive hydrocarbon contaminated aquifers into oxic conditions mainly because of the poor transport properties of oxygen in water (the reason hemoglobin exists in our blood cells is to address that very issue within our bodies). The power of many of the other metabolic systems lies in the fact that they are driven by chemical species that are highly soluble in water (sulfate and nitrate for example). The less efficient energy yield is compensated by the dramatically improved transport properties.
In addition, these alternate metabolic systems are showing to have some capacity to degrade xenobiotic hydrocarbons that prove to be recalcitrant to aerobic attack.
The power of bioremediation lies in harnessing the vitality of bacterial life in the subsurface. The ubiquitous nature of bacterial life is constantly being revised upward. Archaebacteria exist under the extremes of physical/chemical conditions. Bacteria are found at such depths that it is almost certain they were laid down with the surrounding sediments and have been able to survive since. The presence of microbial life on Mars is an even more extreme example of the concept. In-situ bioremediation is a process that appears to be viable almost under any subsurface condition. Our role is to determine the specifics of an appropriate biostimulation program that is not always predicated on an oxygen based system. For some of our most difficult groundwater pollution issues the bacteria are there, waiting.
Return the the Environmental Technology Index Page
Copyright 2002 David B. Vance
All Rights Reserved

If you have comments or suggestions, e-mail me at dbv7@mindspring.com
|