2 The 4 Technology Solutions
![]()
If you have comments or suggestions, e-mail me at dbv7@mindspring.com
MTBE
REALITY AND CONSEQUENCES
An On-Line Version of a Column First Published in:
Environmental Technology Sept./Oct. 1996 Vol. 8 No. 1 Pages 16-17
by: David B. Vance dbv7@mindspring.com
Methyl tert-butyl Ether (MTBE) is on the verge of becoming an issue that could, among other things, cause the reevaluation of the recent trend towards the application of intrinsic remediation [1][2][3][4] to the distal portions of contaminant plumes associated with releases from fuel storage facilities.
MTBE is a key component of modern oxygenated Reformulated Gasoline (RFG). Early use of MTBE as a octane enhancing fuel additive began in the early 1980s. When used as an oxygenation component, summer concentrations are 2% and winter 2.7% (equal respectively to 11% and 15% by volume). The first major use of MTBE to create an "oxygenated fuel" to control air pollution from automobiles occurred in the Denver metropolitan area in 1987. The Clean Air Act of 1990 dictated the adoption of oxygenated fuels within nine major metropolitan areas across the country, full implementation of the plan began 2 years ago in most areas. Currently approximately 36% of all gasoline sold in the United States contains oxygenates, and 85% of those use MTBE. The use of oxygenates is projected to reach 70% within 3 years. However, some areas (such as Milwaukee) have already abandoned the use of MTBE as an oxygenate due to the problems to be discussed.
First it is important to understand the physical properties of MTBE. It is very soluble in water at 4.3%, compared to 0.18% for benzene, 0.05% for toluene, and 0.02% for xylene. This is due to the polar nature imparted to the molecule by the carbon-oxygen-carbon ether bond (see the Figure).
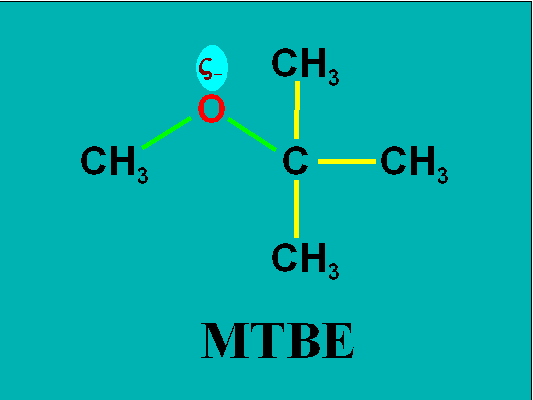
Typical of compounds that have a high degree of solubility in water, the sorption affinity for geologic and carbonaceous components in the soil matrix is very low. Thus MTBE is highly mobile, undergoing no to little retardation as it travels through a groundwater system. Upon release it will rapidly become the lead component of the contaminant plume.
Aside from its high solubility there is a another characteristic of MTBE that contributes to its high mobility, it is extremely resistant to abiotic or biological degradation. The degradation half life of MTBE is poorly and inconsistently documented. Laboratory studies have shown MTBE in groundwater to be recalcitrant to anaerobic [4] as well as aerobic [6] biodegradation, which are the key components of the natural attenuation process that appears to function relatively well and in a geographic sense universally for BTEX hydrocarbons. Field demonstration of MTBE degradation is also inconsistent, potentially presenting the rare circumstance of a compound specific biological degradation capacity being highly variable amongst geographically diverse consortia of similar bacterial species. Isolated instances of MTBE degrading bacteria have been found in chemical plant waste water treatment systems.
The USGS performed a survey of shallow groundwater for MTBE in urban and rural areas in 1995 that serves to illustrate the consequences of the physical character of MTBE. They found that MTBE was present in 27% or the urban wells and 1.3% in rural ones. In addition to point sources such as fuel releases from UST's, it is thought that some portion of the MTBE contamination may be due to volatilization from UST's and Automobiles followed by incorporation into rainfall and infiltration into groundwater systems. The recalcitrant MTBE molecule does not degrade well in the presence of open air and sunlight.
Given what appears to be an increasing presence in groundwater, the issue then becomes one of toxicity. Originally it was thought that MTBE was relatively non-toxic. That, its ease of manufacture, and low cost are the prime reasons it was selected by the EPA for RFG use in the 1990 Clean Air Act. However, public complaints rapidly followed the use of MTBE in a number of diverse geographic areas. As a result, studies were initiated to further evaluate the toxicity of MTBE. There are preliminary indications that at high concentrations it may be a carcinogen. Metabolic and oxidation daughter products of MTBE include tert-butyl formate and formaldehyde, are both classified by the EPA as probable carcinogens. It is likely to prove that low concentrations of MTBE are not highly toxic (except to perhaps a small portion of the population with unusual chemical sensitivity). However, the worst of all the problems associated with the public and MTBE impact are the levels at which the human senses can detect it, 15 to 95 ppb for smell and 39 to 134 ppb for taste. It smells like turpentine. With regards to the public acceptance of risk, perception is paramount. Consequently, the EPA has proposed a health advisory level of 70 ppb. In California the detection limit for purposes of reporting is 5 ppb.
One positive aspect of the physical character of MTBE is that removal from a groundwater system via pump and treat requires significantly fewer pumped volumes compared to BTEX hydrocarbons. In fact, pump and treat is to date the only viable in-situ treatment option for MTBE contamination. However, its chemical recalcitrance also impacts surface based treatment systems. Activated carbon will work, although poorly (read expensive). Air stripping can work, but poorly. Lastly are chemical oxidation systems such as ozonization which to date are uncertain with regards to effectiveness as well as the generation of toxic daughter oxidation products. It is estimated that the treatment of water impacted with dissolved MTBE is thirty times more expensive than treatment of an analogous stream containing BTEX.
The consequences are just beginning to be felt. In October of 1997 Tosco Corporation (the third largest refiner in California) called for the phase out of MTBE, in December Chevron and others appealed to the US Congress and California regulators to allow the production of cleaner burning gasoline’s without the use of oxygenates such as MTBE. All are concerned about liability. Tremendous sums of money are at stake, MTBE is third in production of all organic chemicals manufactured in the United States (17.6 billion pounds in 1995), the agricultural industry is waiting in the wings with ethanol, and refiners have the technology to produce cleaner fuels through innovative refinery processes.
Meanwhile, the proverbial horse may be out of the barn, just as regulatory agencies and polluters were beginning to get comfortable with the concept of intrinsic remediation, MTBE could present a problem that may obviate that approach.
Return to the Environmental Technology Index Page
Copyright 2008 David B. Vance
All Rights Reserved![]()
If you have comments or suggestions, e-mail me at dbv7@mindspring.com