|
2 The 4 Technology Solutions

TRANSVERSE DISPERSION DRIVES NATURAL ATTENUATION
An On-Line Version of a Column First Published in:
Environmental Technology Jan./Feb. 1997 Vol. 7 No. 1 Pages 31-32
by: David B. Vance dbv7@mindspring.com
In our last column we discussed how groundwater flow stimulated by pump and treat systems can act as a source of electron acceptors for the natural attenuation of contaminating hydrocarbons. This column will examine the effect of dispersion on that process in groundwater systems under natural conditions.
Dispersion is the process by which the interface of contaminated groundwater with native groundwater does not remain abrupt. The leading edge of a contaminant plume will arrive at a given point more rapidly than it would if advection alone where the acting driving force. The mean transport velocity of the contaminant mass remains the same, but concentration gradients are set-up. This occurs simultaneously with the phenomena of the contaminant occupying, with time, an increasing volume of groundwater.
There are two fundamental types of dispersion, longitudinal and transverse. Both are accentuated by the inhomogenous and anisotropic physical configuration (1)(2) of the permeable matrix within a groundwater system.
- Longtitudinal dispersion is caused by differences in groundwater velocity through pore spaces that vary in width or tortuosity. The result is dispersion that occurs along the direction of groundwater flow.
- Transverse dispersion is driven by groundwater flowing around individual particles in the aquifer matrix, the effect occurs perpendicular to the groundwater flow direction.
Transverse dispersion is effective from the edges of an elongated plume and occurs only when there is a point source of contamination. In the context of natural attenuation Longtitudinal dispersion is purely a dilution phenomena.
Both types of dispersion are dependent upon horizontal and vertical variations in permeability. Increasing anisotropy and heterogeneity increase the magnitude of dispersion. Groundwater velocity also plays a role, at low velocities the effects of diffusion will equal those of dispersion.
- The ratio of longitudinal to transverse dispersivity ranges from 1 to 24,
- most commonly horizontal transverse dispersivity is 20% to 10% of the longtitudinal dispersivity, and
- vertical transverse Dispersivity is 2% to 1% of the longtitudinal dispersivity.
Instead of "football" shaped plumes, this difference in vertical and horizontal dispersivity tends to generate plumes that in three dimensions are "surfboard" shaped. The determination of the specifics of the effect of dispersivity on a contaminant concentration at a given time and location in an aquifer is an extremely complex process that requires:
- detailed knowledge of the physical configuration of the aquifer matrix and
- solution to partial differential equations for final values.
- The physical scale that is examined also has impact, values of dispersivity change as one examines an aquifer on the scale of inches, feet, or thousands of feet.
For purposes of this column we are largely concerned over the effect of transverse dispersion. That is what serves to physically mix groundwater containing contaminants with adjacent groundwater that is unimpacted and contains natural electron acceptors.
The phenomena is probably best understood in terms of angle of divergence, that is the angle between the two edges of the plume as it migrates from a point source.
- In granular materials that angle can be as low as 2 degrees, and
- in fractured aquifers as high as 20 degrees.
- The most accurate determination of the angle of divergence is from information gathered as close as possible to the point representing the source of contamination.
The picture we normally have of a contaminant plume as it migrates away from a point source is one that is (in the two horizontal dimensions) tear drop shaped.
- The plume initially spreads downgradient and cross gradient until some point is reached at which
- the distal edges of the plume first travel parallel to the advective groundwater flow, and
- then begin to turn inward to the plume axis and close.
The distal portion of the plume is the dominant area where longitudinal dispersion (as well as transverse dispersion) is in effect. Adsorption and other attenuation reactions also take place, but for the purposes of this discussion those effects are ignored. Under pure hydrodynamic effects a plume should dilute itself taking the shape of a cone at a constant angle of dispersion. This assumes that the nature of the geologic matrix and groundwater velocity stays constant. which it probably will not (particularly vertically).
The point is to realize is that the downgradient shape of the plume is a consequence of attenuation caused by the migration of electron acceptors into the margins of the plume through the force of transverse dispersion. Not through purely hydrodynamic flow effects.
Now, to the core of this column. Figure 1 illustrates the groundwater mixing velocity of transverse dispersion over the range of 2 to 20 degrees.
- The X axis is aquifer groundwater velocity in feet per day, this is the natural groundwater velocity with no pumping system operating.
- The Y axis is the mixing velocity due to the effect of transverse dispersion.
- This velocity has 2 components,
- one is the force driving the contaminant plume outward into the surrounding aquifer and
- the second is driving uncontaminated groundwater into the plume.
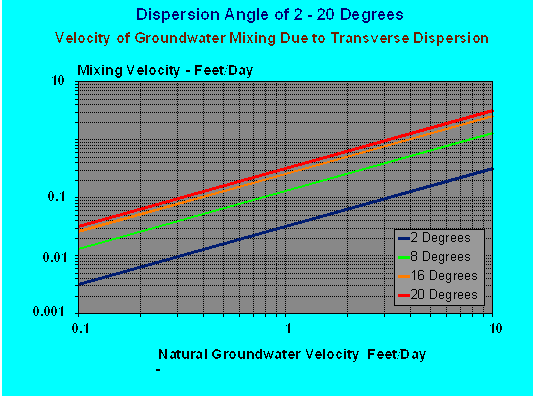
Figure 1 - Effect of Dispersion Angle of Plume on Mixing Velocity
Figure 2 converts mixing velocity into degradation rates of dissolved hydrocarbon contamination with a number of different electron acceptors. Please review last issues column for more details on the specifics of the chemistry of those processes.
To determine degradation times for adsorbed hydrocarbons, half the mixing velocity determined from Figure 1 and use that velocity value for groundwater velocity presented in the Figure presented in last issues column.

Figure 2 - Effect of Mixing Velocity on Degradation Rates
This information serves to provide an estimate of time frames required for the various natural attenuation mechanisms to be effective. A key component of this process is the removal of the source of further contamination. Under many conditions time frames are slow, but adequate for dissolved and low level adsorbed contamination. These rates are not adequate for remediation of source areas.
Return to the Environmental Technology Index Page
Copyright 2008 David B. Vance
All Rights Reserved

If you have comments or suggestions, e-mail me at dbv7@mindspring.com
|